Gadolinium-based contrast agents (GBCAs) have been used routinely in neuroradiology since their introduction in the late 1980s and provide invaluable clinical information for the diagnosis and management of patients with neurologic diseases. These agents have been generally considered safe for patients with normal renal function, with over 450 million doses administered worldwide.
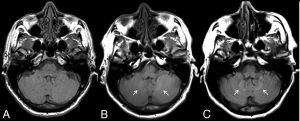
In 2014, Kanda et al1 reported an association between the number of prior GBCA administrations and the development of hyperintense signal in the globus pallidus and dentate nucleus on unenhanced T1-weighted images. Shortly afterwards, McDonald et al2 confirmed the presence of gadolinium within brain tissue in adult decedents who had undergone GBCA-enhanced MRI. We have shown on MRI and at autopsy that gadolinium is also retained within the pediatric brain.3–5
GBCAs can be classified as either macrocyclic or linear based on the structure of the gadolinium chelate. Several studies, in both adult and pediatric populations, have evaluated various GBCAs with the general consensus being that linear, but not macrocyclic, GBCAs are associated with long-term gadolinium retention in the brain. In our study, we found the development of high signal intensity within the globus pallidus and dentate nucleus following multiple administrations of the linear agent gadobenate dimeglumine.6
Currently, there is little scientific evidence linking gadolinium deposition in the brain with specific clinical symptoms, though this is an area of active research. Our group in particular is studying gadolinium deposition in pediatric patients given the unique vulnerabilities of children, including ongoing central nervous system development, immature renal function, and active bone formation. In adults, gadolinium is known to accumulate in bone tissue at levels approximately 23 times higher than those in the brain.7 It is unknown whether rapid bone turnover due to growth and development enhances gadolinium accumulation in children. To investigate this, we are currently measuring gadolinium levels in sternal bone in children who have undergone cardiac surgery. Results from this study will be important in determining whether bone represents a long-term storage site for gadolinium in children. If so, patients receiving large cumulative GBCA doses during childhood may be at risk for secondary endogenous gadolinium exposure much later in life (eg, during periods of bone loss).
Given the current uncertainties concerning the clinical significance of gadolinium deposition within the body, many practices have chosen to take a cautious stance and have switched to the exclusive use of higher-stability, macrocyclic agents.8 In 2016, the American College of Radiology and the American Society of Neuroradiology issued a joint position statement recognizing the propensity of GBCAs to deposit in sensitive tissues such as the brain.9 In July 2017, the Pharmacovigilance Risk Assessment Committee of the European Medicines Agency recommended suspension of the marketing authorizations for 4 linear GBCAs.10 The U.S. Food and Drug